Aluminium reacting with bromine
Numéro d’image : 13619261
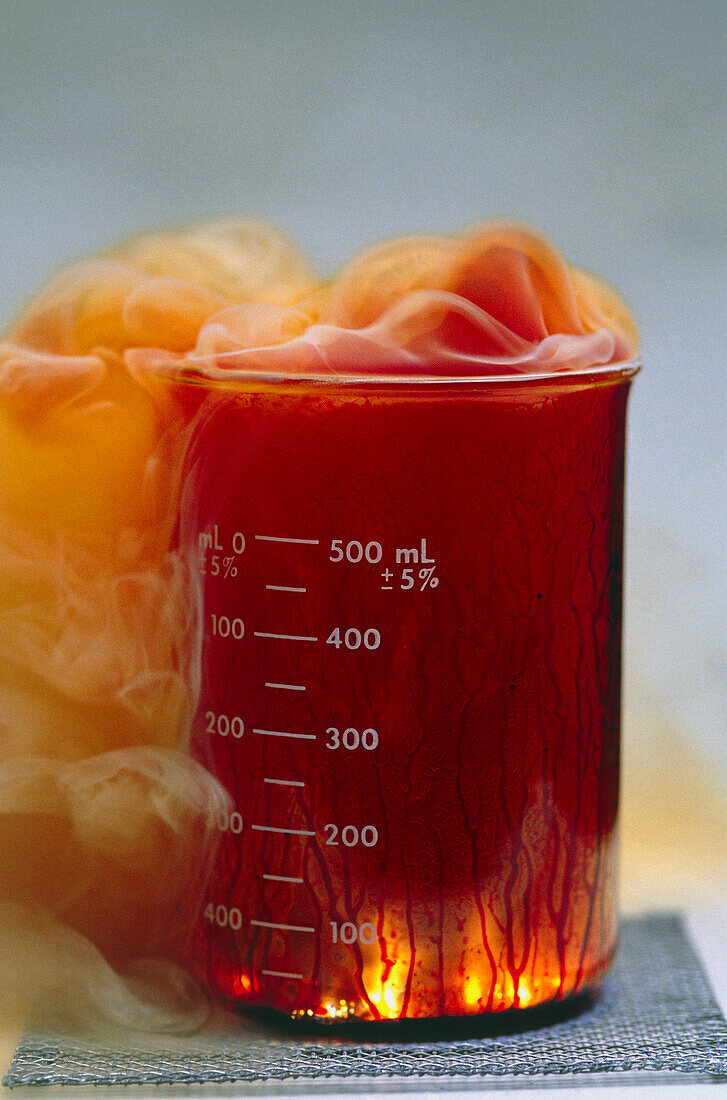
| Aluminium reacting with bromine. Aluminium metal (Al) burning in a beaker full of the halogen bromine (Br2) to produce solid aluminium(III) bromide, AlBr3. The equation for this reaction is: 2Al + 3Br2 ---> Al2Br6 Aluminium bromide is used as a catalyst in organic chemistry. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Winters, Charles D. |
| Taille de l’image : | 3516 px × 5322 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
