Model of ethene before hybridisation LHS
Numéro d’image : 13417303
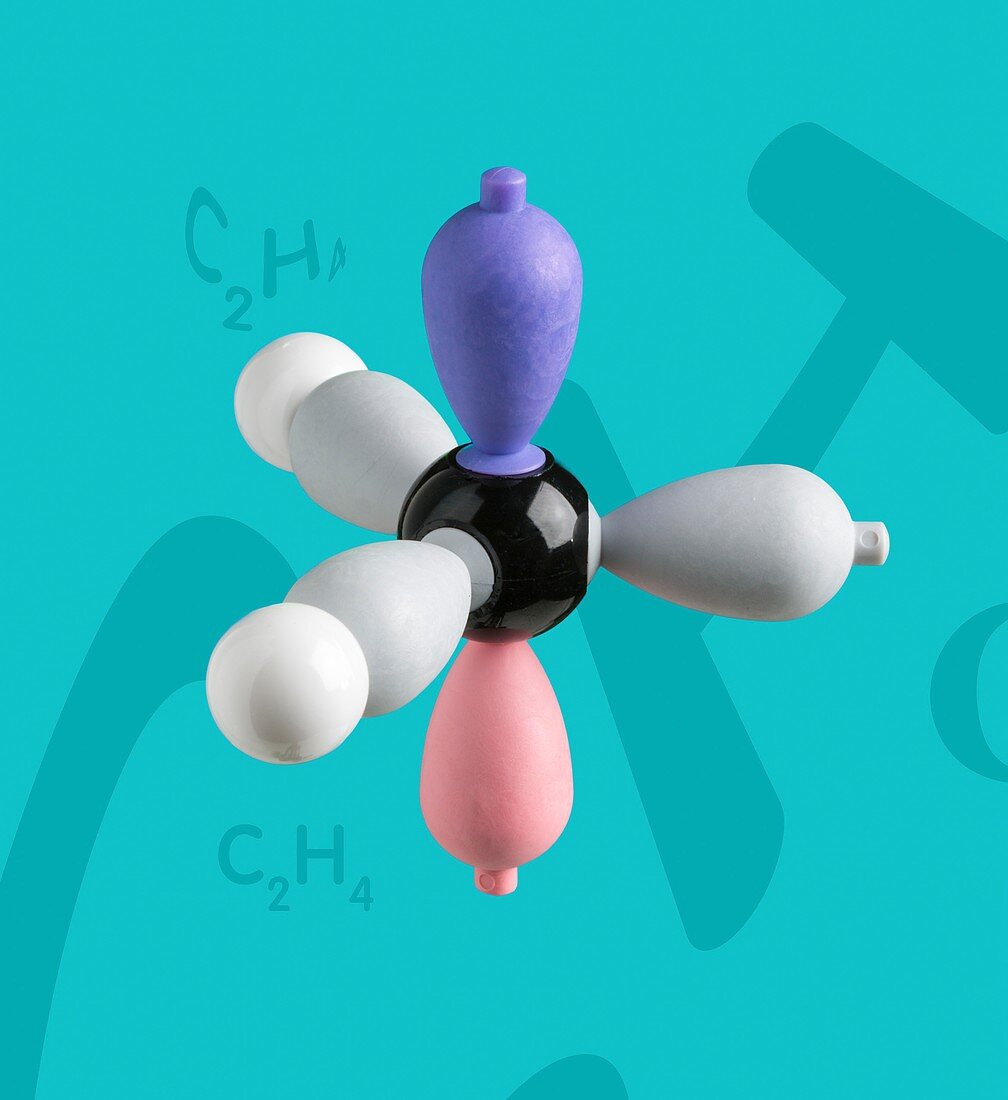
| Electron density model of ethene before hybridisation Left hand side of Ethene model before hybridization. Ethene is a very important hydrocarbon which has the formula C2H4. The model shows sigma- and pi-bonding orbitals, and the concept of hybridisation and delocalisation. A carbon and two hydrogen atoms form the left and right sides of the simplest alkene compound - ethene. In an sp2 hybridization, one s orbital is mixed with two p orbitals to form three sp2 hybridized orbitals. Much of the production of ethene goes to polyethylene manufacture and also as a plant hormone to speed up the ripening of fruit. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Chillmaid, Martyn F. |
| Taille de l’image : | 4001 px × 4368 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
