Copper penny reacts with nitric acid
Numéro d’image : 12653515
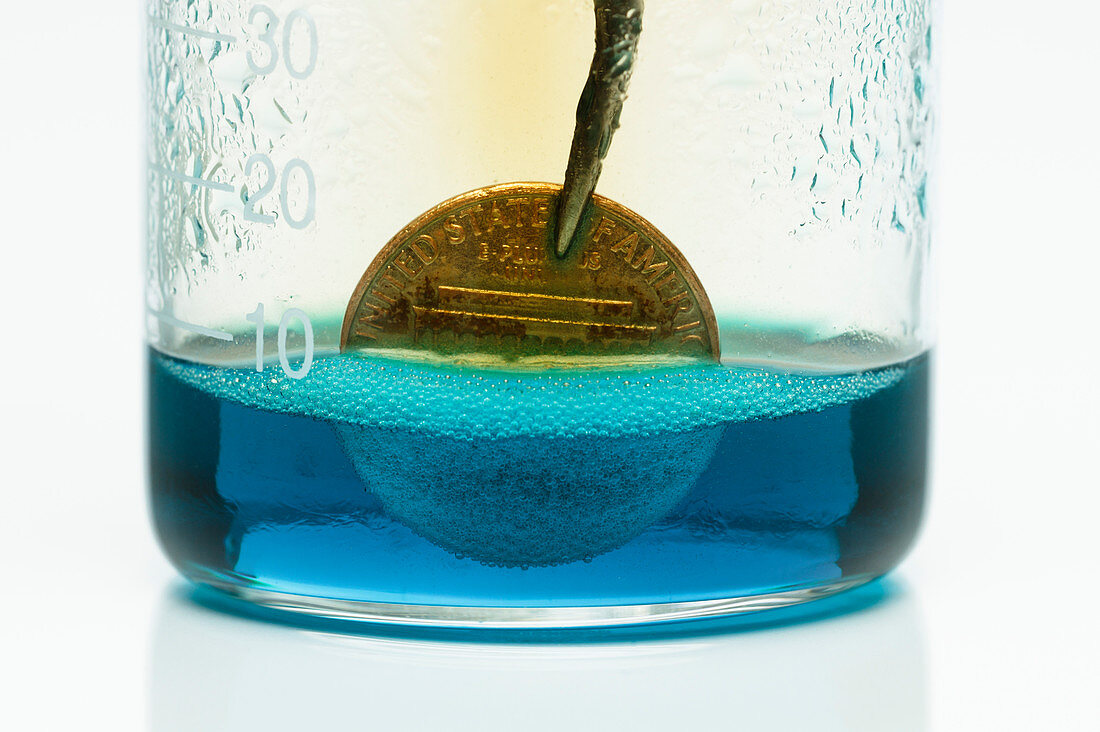
| Copper penny reacts with nitric acid. US copper penny (1 cent coin minted in 1980) is partially immersed into concentrated (8M) nitric acid (HNO3). Copper is oxidized to form blue copper nitrate (Cu(NO3)2) and brown nitrogen dioxide gas (NO2) bubbles: Cu + HNO3 -> Cu(NO3)2 + NO2 + H2O. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Science Source / Turtle Rock Scientific |
| Taille de l’image : | 5152 px × 3429 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
