Reaction Rate
Numéro d’image : 12636511
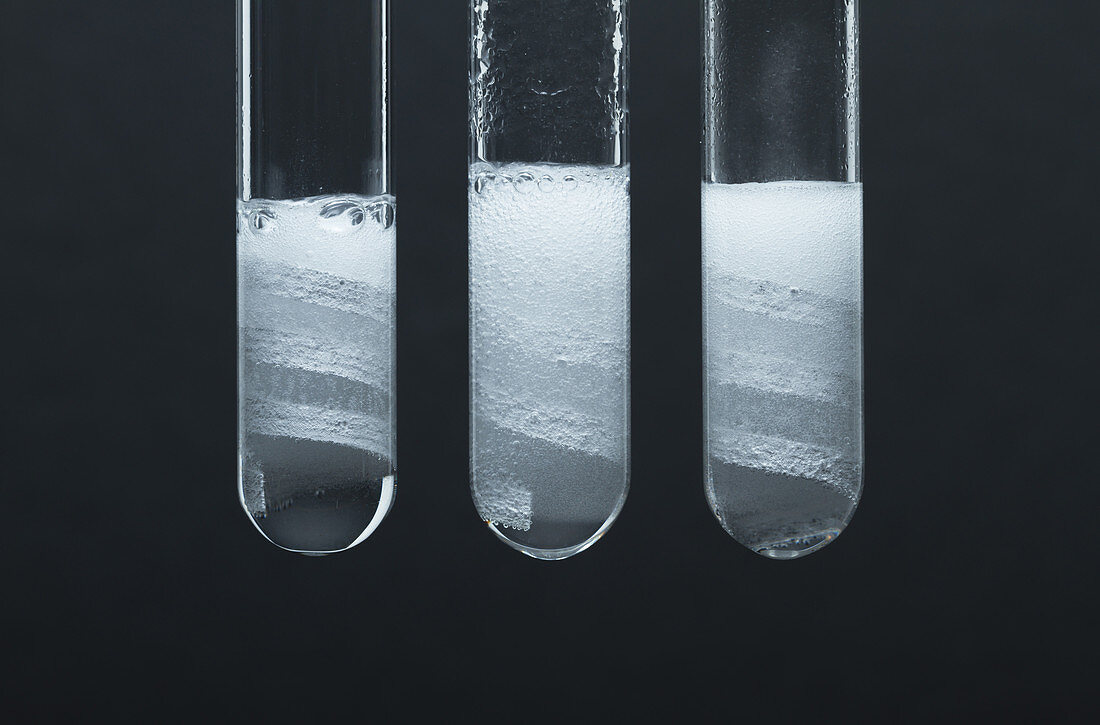
| Reaction rate. Magnesium coils are placed in test tubes with left: concentrated (6M) acetic acid (CH3COOH); centre: diluted (1M) hydrochloric acid (HCl); and right: diluted (1M) acetic acid. Magnesium reacts with the acids producing hydrogen bubbles: Mg + 2H+ -> Mg+ + H2. Hydrochloric acid is a strong acid and completely dissociates in solution. Acetic acid is a weak acid and only partially dissociates. The reaction proceeds with about the same rate in concentrated acetic acid and diluted hydrochloric acid (judged by the size and rate of hydrogen bubbles production). The reaction is slower in a diluted acetic acid. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Giphotostock |
| Taille de l’image : | 5131 px × 3383 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
Mots clés
- acide,
- acide acétique,
- acide chlorhydrique,
- alcalin,
- bulles,
- CH3COOH,
- chimie,
- chimique,
- chlorure de magnésium,
- concentration,
- éprouvette,
- H2,
- hydrogène,
- leucémie à tricholeucocytes,
- magnesium,
- magnésium,
- métal,
- métal alcalino-terreux,
- métal de la terre,
- métal de terre,
- Mg,
- mycoplasma genitalium,
- pétiller,
- réaction,
- réaction de déplacement,
- réactivité,
- seul,
- taux,
- taux de réaction,
- unique,
- vitesse de réaction
