Acid-base Titration
Numéro d’image : 12629778
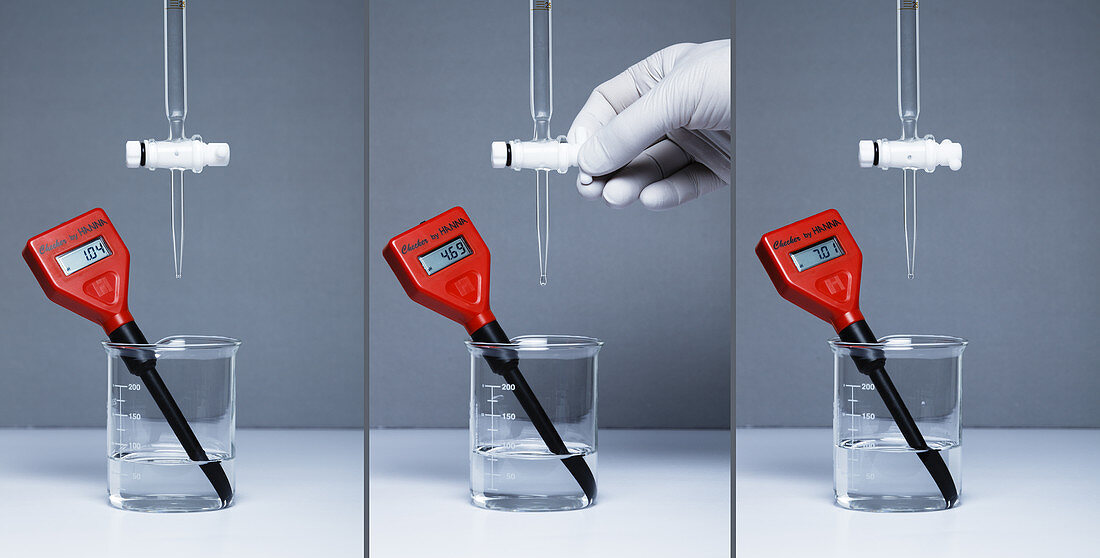
| Acid-base titration. A pH meter is used in an acid-base titration. Here a beaker contains a strongly acidic solution, pH=1, while a burette above it contains a base with a known concentration (left frame). As the base is added to the solution, its pH increases (centre frame). When the pH reaches 7, i.e. the solution becomes neutral (right frame), the volume of the base used is recorded and the initial concentration of the acidic solution (or the number of moles of H+ ions) can be calculated. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Giphotostock |
| Taille de l’image : | 7092 px × 3600 px |
| Model Release : | Non requis |
| Restrictions : |
|
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
Mots clés
- acide,
- acide base,
- acide-base,
- acidité,
- acido-basique,
- alcalin,
- alcalinité,
- burette,
- chimie,
- chimique,
- compte-gouttes,
- erlen,
- erlenmeyer,
- fiole,
- fiole Erlenmeyrer,
- main gantée,
- mesurant,
- mesure,
- mesurer,
- neutralisation,
- neutraliser,
- PH,
- Ph-mètre,
- pipette,
- réaction,
- séquence,
- séries,
- solution,
- titrage
