Hot ice, molecular model
Numéro d’image : 12581245
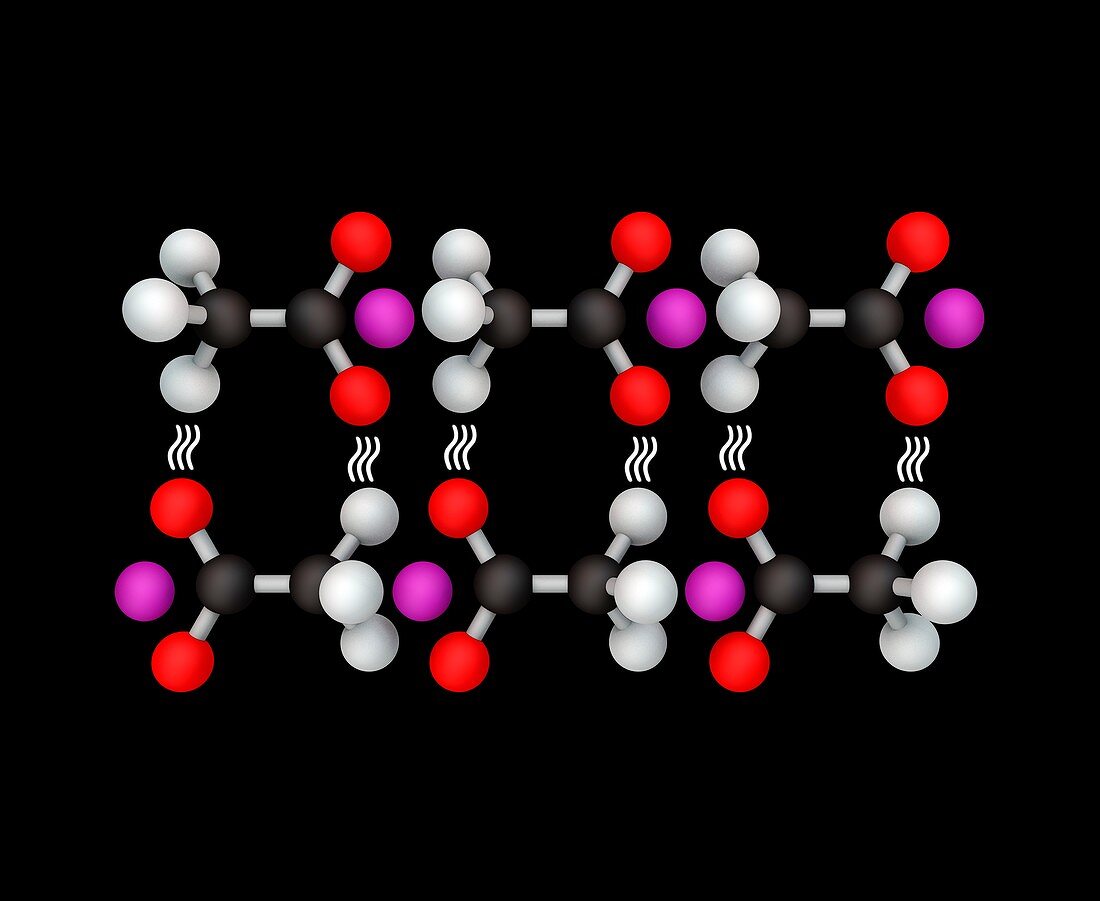
| Hot ice, molecular model. Hot ice is formed when a solution of sodium acetate is cooled below its melting point, causing crystallisation. This is an exothermic, or heat-producing, reaction, so the resultant clear crystal is warm. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Jensen, Mikkel Juul |
| Taille de l’image : | 6532 px × 5350 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
