Diamond crystal structure
Numéro d’image : 12445464
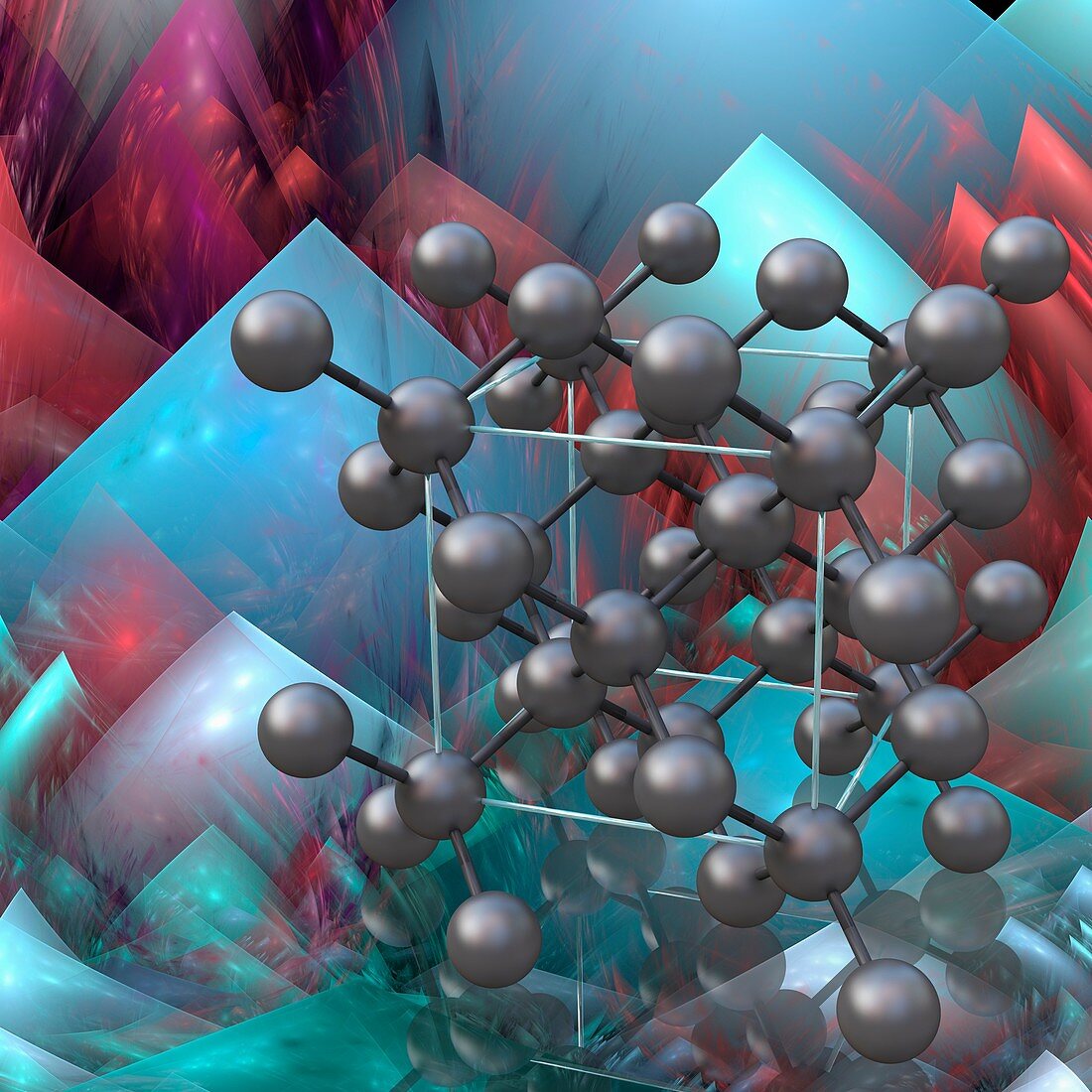
| Diamond. Computer illustration showing the molecular structure of diamond, a form of the element carbon. Carbon atoms are shown as spheres, linked by rigid bonds. Each carbon atom in a diamond molecule is positioned at the centre of a tetrahedron, linked to four other atoms at the corners of the tetrahedron by strong covalent bonds. This repeating tetrahedral arrangement allows no rotation about the bonds, and so the structure is completely rigid, making diamond the hardest known naturally-occurring material. It is used in industry to tip heavy-duty cutting and drilling equipment and is also a precious gemstone. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Laguna Design |
| Taille de l’image : | 4180 px × 4180 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
Mots clés
- ADN,
- atome,
- atomes,
- aucun,
- carbone,
- chimie,
- chimique,
- collage,
- composé,
- composés,
- cristal,
- diamant,
- éléments,
- gemme,
- illustration,
- le plus dur,
- liaison,
- liaison covalente,
- liaisons,
- liens,
- modèle,
- moléculaire,
- molécule,
- obligations,
- oeuvre,
- organique,
- personne,
- pierre précieuse,
- structure,
- tétraédrique
