Hydrogen peroxide decomposition
Numéro d’image : 11997202
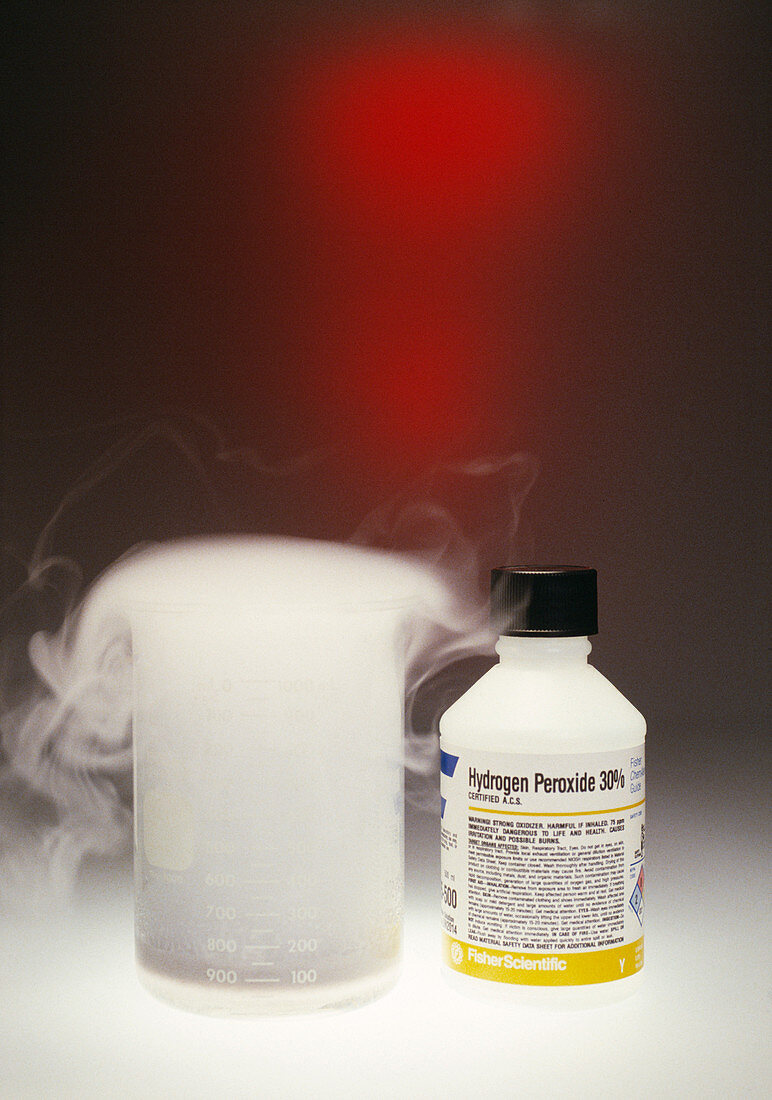
| Hydrogen peroxide decomposition. Demonstration of hydrogen peroxide decomposition being catalysed by manganese(V) oxide. The beaker at left contains hydrogen peroxide,which is breaking down into oxygen (seen as white vapour) and water. Manganese(V) oxide is said to be a catalyst because it increases the rate of this reaction without itself reacting or changing. The equation for this reaction is: 2H202 ----> 2H20 + O2 Hydrogen peroxide is a powerful oxidizer. It is used as a bleach for clothes,textiles,and paper products. It is also used in chemical engineering and in pollution control | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Winters, Charles D. |
| Taille de l’image : | 3142 px × 4475 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : |
|
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
