Double exposure of corrosion on steel and copper
Numéro d’image : 11899619
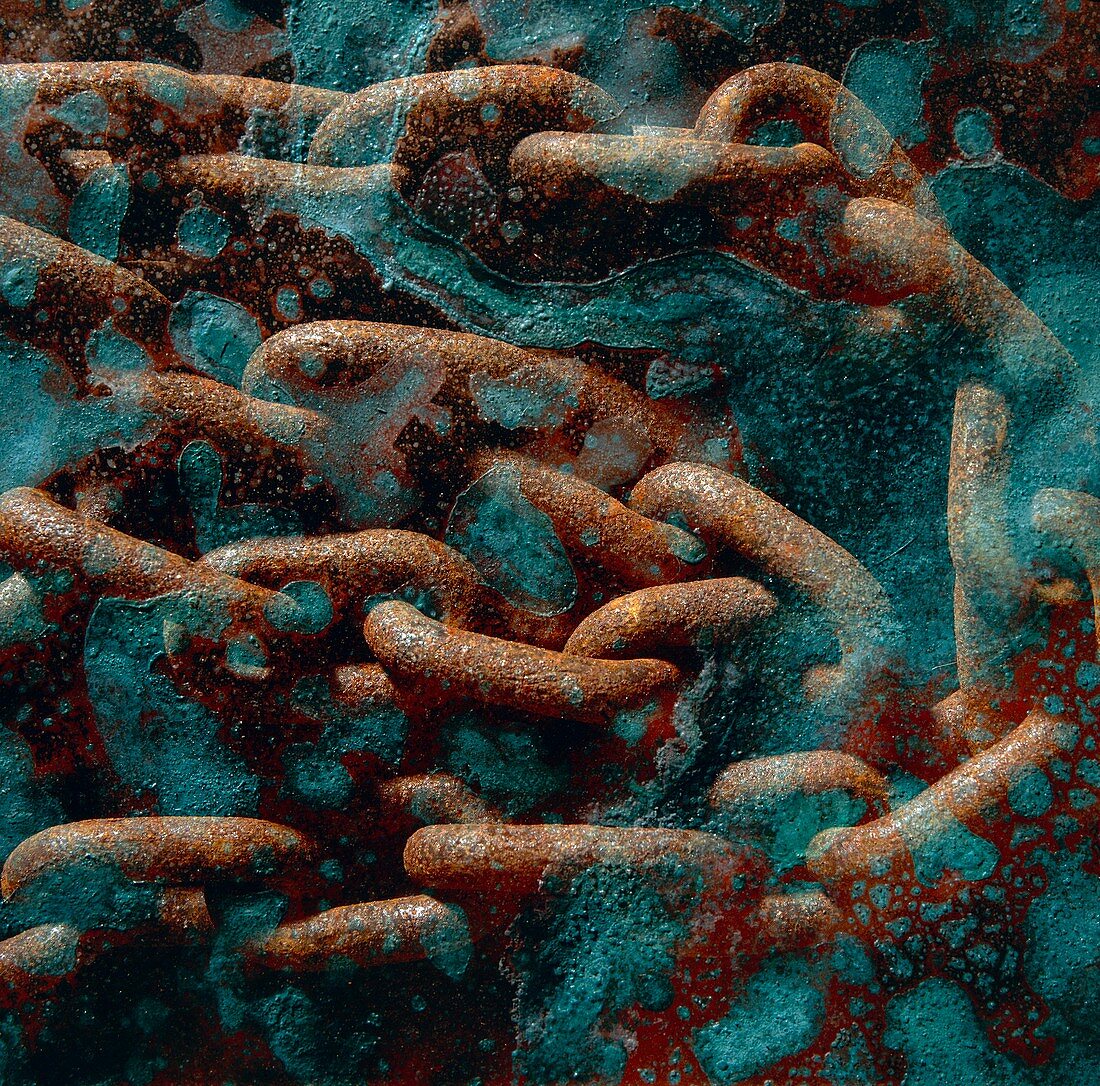
| Corrosion. Double exposure showing the effect of corrosion on copper and on a steel chain. Copper is a yellow-red,soft,very malleable and ductile metal. It is unattacked by dry air but in moist air containing carbon dioxide (CO2) a green film of basic carbonate is formed. Rust is seen on the steel chain. It consists in the formation of a loose,flaky layer of hydrated iron oxide on the surface of iron or steel. The reaction proceeds in a moist atmosphere and it is accelerated by the presence in the atmosphere of sulphur dioxide,sulphuric acid and chlorine ions. Rusting is negligible in dry,hot areas as the critical humidity is too low | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / De Schwanberg, Victor |
| Taille de l’image : | 4250 px × 4196 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
