Cyclohexane molecule (boat form)
Numéro d’image : 11523048
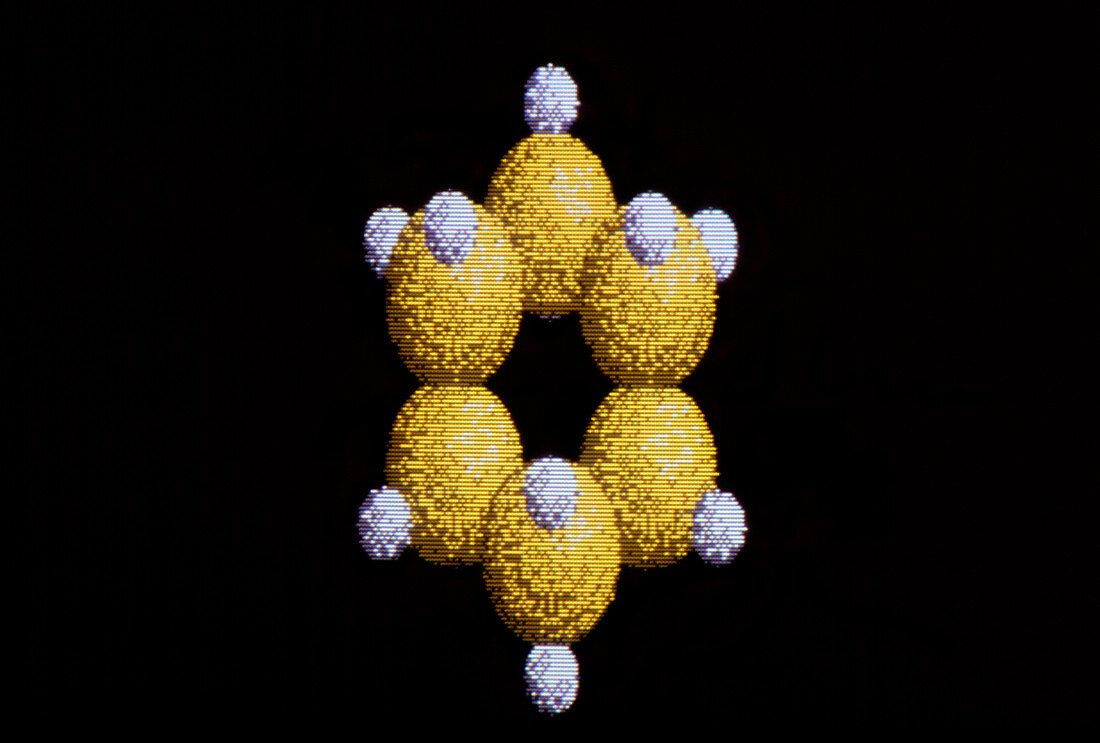
| Cyclohexane. Computer graphics representation of a molecule of cyclohexane (chemical formula C6H12),shown in its chair conformational form. Six carbon atoms (yellow) are arranged hexagonally on various planes; hydrogen atoms are white. A planar hexagon,however,would require a carbon-carbon (C-C) bond angle of 120 degrees,resulting in a highly strained form. Through rotation about the C-C bond,cyclohexane is able to exist in two "puckered" forms which are completely free from strain,known as chair and boat conformations. Cyclohexane exists pre- dominantly in the chair form as this corresponds to a lower (more stable) energy value. Compare A705/016 showing boat form | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Kulyk, Mehau |
| Taille de l’image : | 3726 px × 2516 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
