Odour molecule vibration
Numéro d’image : 11522762
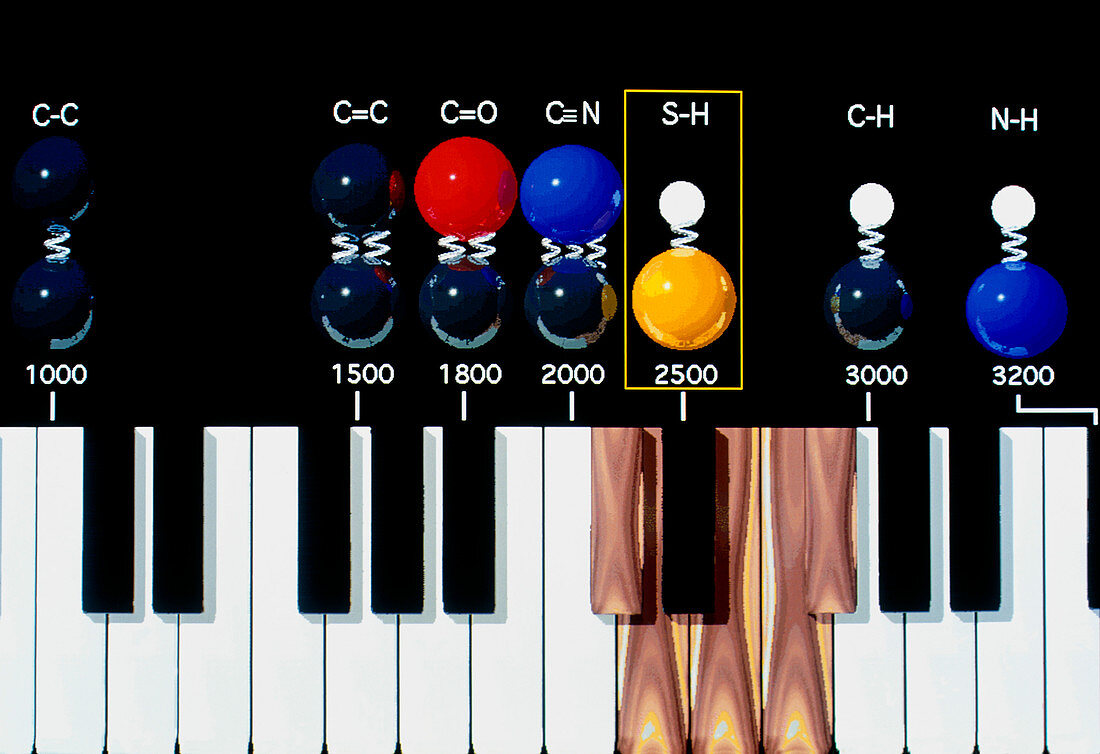
| Odour molecule vibration. Abstract computer art- work of vibrating molecular bonds. Bond vibration is the main factor which determines how a molecule smells. The bonds are shown on a keyboard,& their constituent atoms are colour-coded: carbon (C) is dark blue,oxygen (O) is red,nitrogen (N) is light blue,sulphur (S) is yellow and hydrogen (H) is white. The numbers beneath each bond correspond to its vibrational wavenumber - the number of wavelengths of infrared light at that energy which would fit in a centimetre. The S-H bond is high- lighted because very little else vibrates in the 23-2700 wavenumber region,giving molecules which contain the bond a distinctive sulphuraceous smell | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Parker, David |
| Taille de l’image : | 4993 px × 3423 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
