ATP bind site of phospho- glycerate kinase
Numéro d’image : 11520935
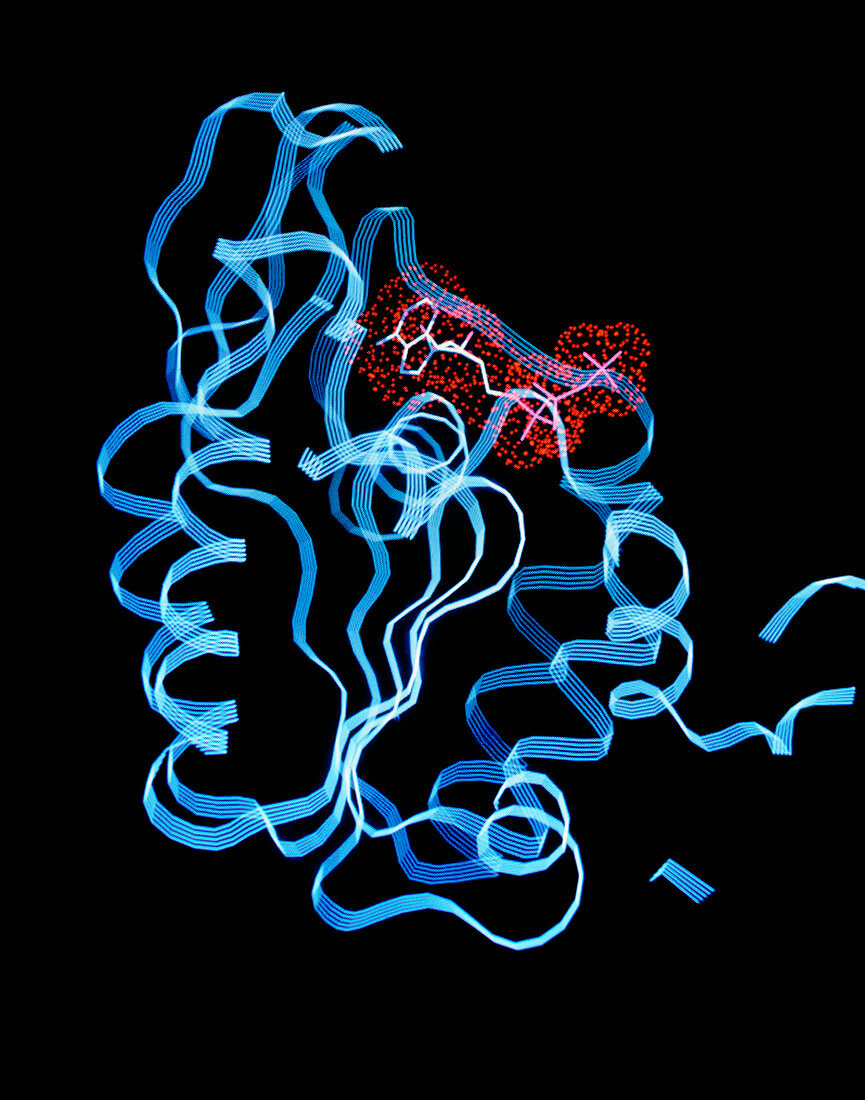
| Computer graphics representation of the ATP- binding domain (reaction site) of phosphoglycerate kinase,a glycolytic enzyme (catalyst). In glycolysis,glucose is converted to pyruvate with the production of ATP: in other words,a catabolic reaction provides metabolic energy (as ATP) at the expense of the degradation of food molecules (glucose). This crystal structure is of horse phosphoglycerate kinase,a bi-lobed structure composed of two domains (one for each reactant),each composed of a six-stranded beta sheet surrounded by alpha helices (blue ribbons). Here,Van der Waals bonded ATP appears in red. This enzyme catalyses the transfer of a phosphoryl group from 1,3-diphosphoglyerate to ADP to yield ATP & 3- phosphoglycerate. Proposed mechanism involves hinge- bending to unite substrates | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / OXFORD MOLECULAR BIOPHYSICS LABORATORY |
| Taille de l’image : | 4172 px × 5305 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
