Model of sodium chloride crystal lattice
Numéro d’image : 11520287
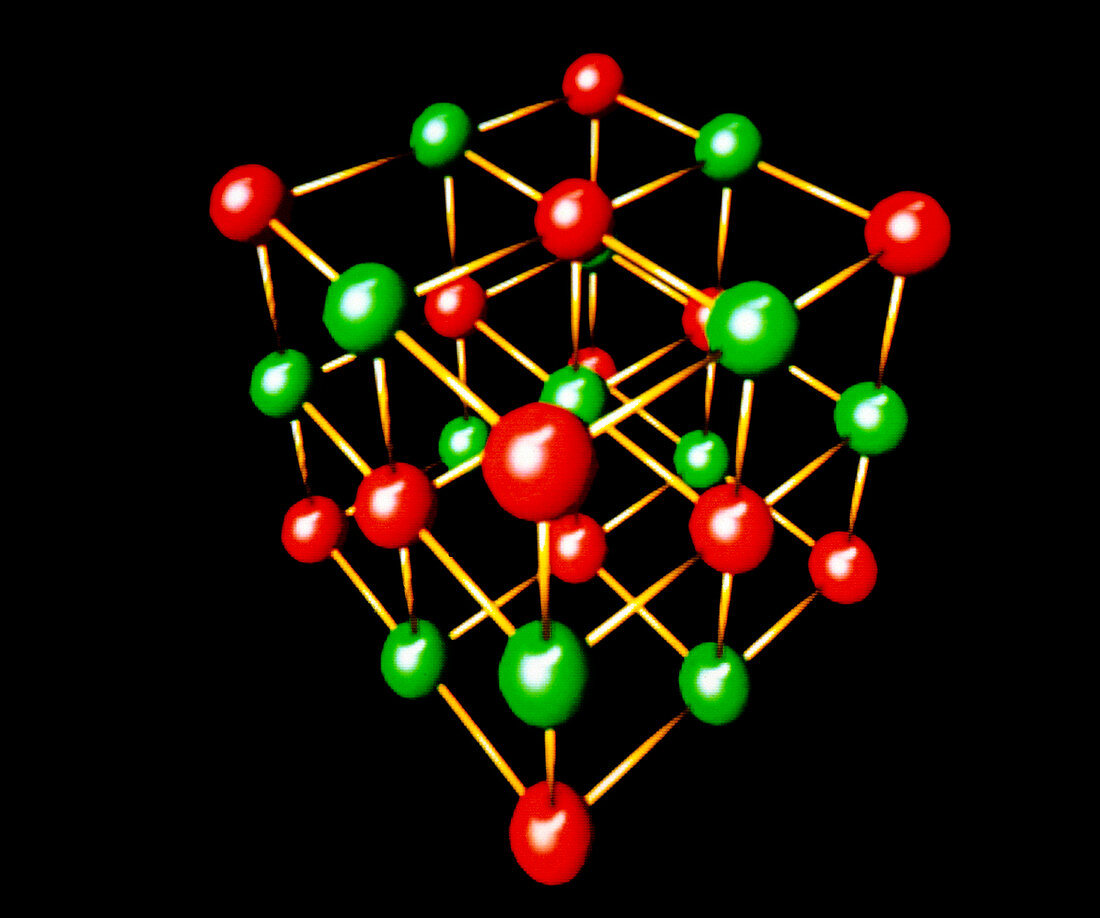
| Model of the cubic crystal lattice of sodium chloride (pure common salt). The cubic crystal consists of an ionic lattice of Sodium (Na+) and Chlorine (Cl-) ions,where the repeating units of the lattice are positively & negatively charged respectively. Sodium ions are represented by red spheres,chlorine ions as green spheres. Ionic crystals tend to be hard & brittle,due to the strong electrostatic forces between the constitutent ions | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Pasieka, Alfred |
| Taille de l’image : | 3543 px × 2956 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
