Limestone reacting with acid
Numéro d’image : 11519885
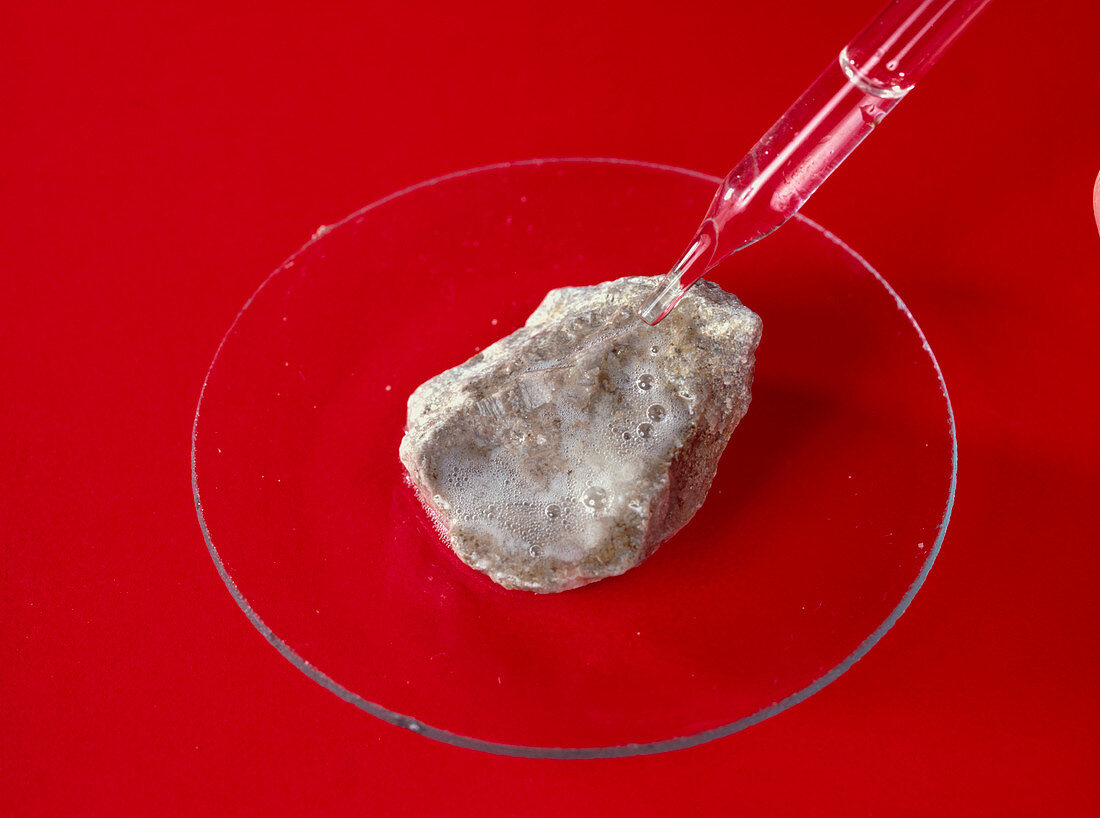
| Limestone reacting with acid. Pipette being used to add dilute acid to a lump of limestone (calcium carbonate,CaCO3). Bubbles of carbon dioxide gas are being produced in the resulting reaction. This is an acid-base reaction,where calcium carbonate (the base) reacts with the acidic hydrogen (H+) to form aqueous calcium ions & carbonic acid (H2CO3). This acid is unstable,and rapidly decomposes to form carbon dioxide (CO2) and water (H2O). This is how rainwater erodes limestone. The equations for this reaction are: 1) CaCO3 + 2H(+) ----> Ca(2+) + H2CO3 2) H2CO3 ----> CO2 + H2O | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Andrew Lambert Photography |
| Taille de l’image : | 3994 px × 2970 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
Mots clés
- acide,
- apparatus,
- appareil,
- base,
- bulle,
- bulles,
- calcaire,
- carbonate de calcium,
- chimie,
- chimique,
- chuter,
- démonstration,
- démontrer,
- déposer,
- dilué,
- dioxyde de carbone,
- éroder,
- érosion,
- expérience,
- gaz,
- manifestation,
- manifester,
- morceau,
- pétiller,
- pipette,
- pluie acide,
- produits chimiques,
- réaction,
- réagir,
- roche,
- tomber,
- verre à montre,
- verre de montre,
- verrerie
