Oxidation of alcohols
Numéro d’image : 11519867
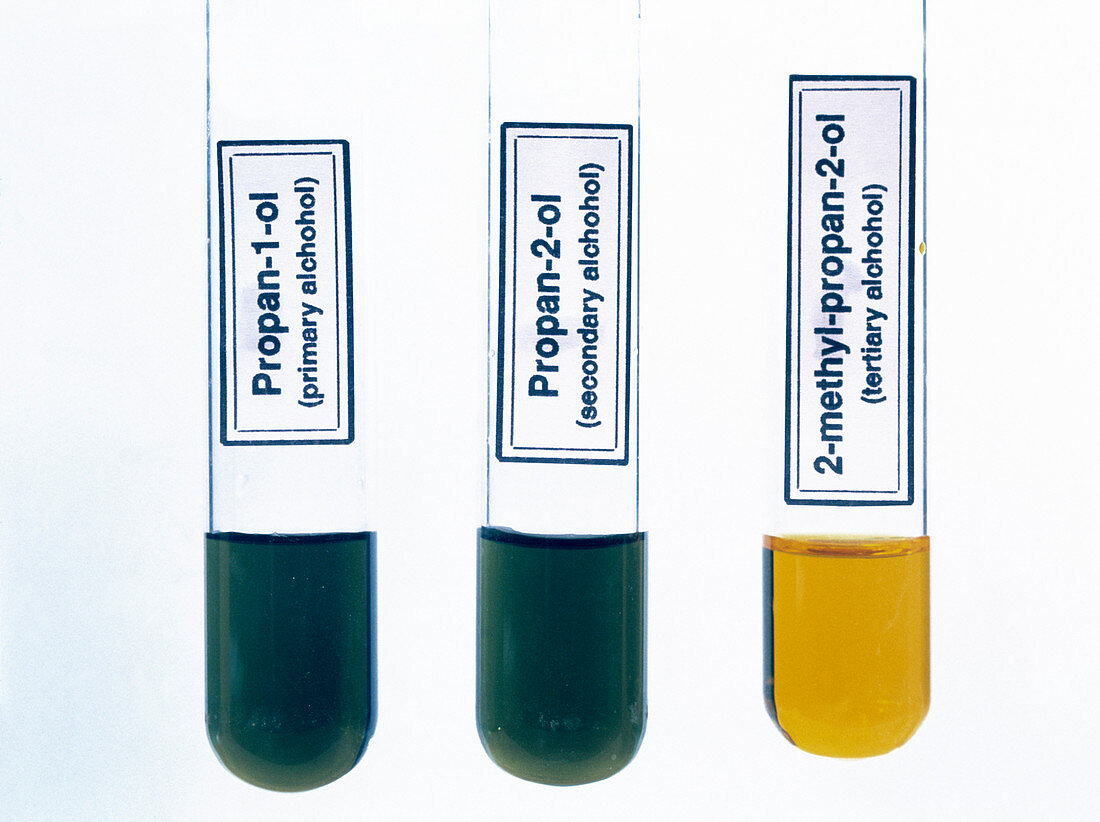
| Oxidation of alcohols. A strong oxidizing agent (potassium dichromate,K2Cr2O7,yellow) has been added to three alcohols. A reaction has occurred with the primary and secondary alcohols (left & centre). The tertiary alcohol (right) has not reacted. A primary or secondary alcohol has a hydroxyl group (-OH) attached to a carbon atom joined to two or one hydrogen atoms respectively. A tertiary alcohol has a hydroxyl group attached to a carbon with no hydrogen atoms | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Andrew Lambert Photography |
| Taille de l’image : | 4356 px × 3257 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
