Ammonium chloride formation
Numéro d’image : 13361098
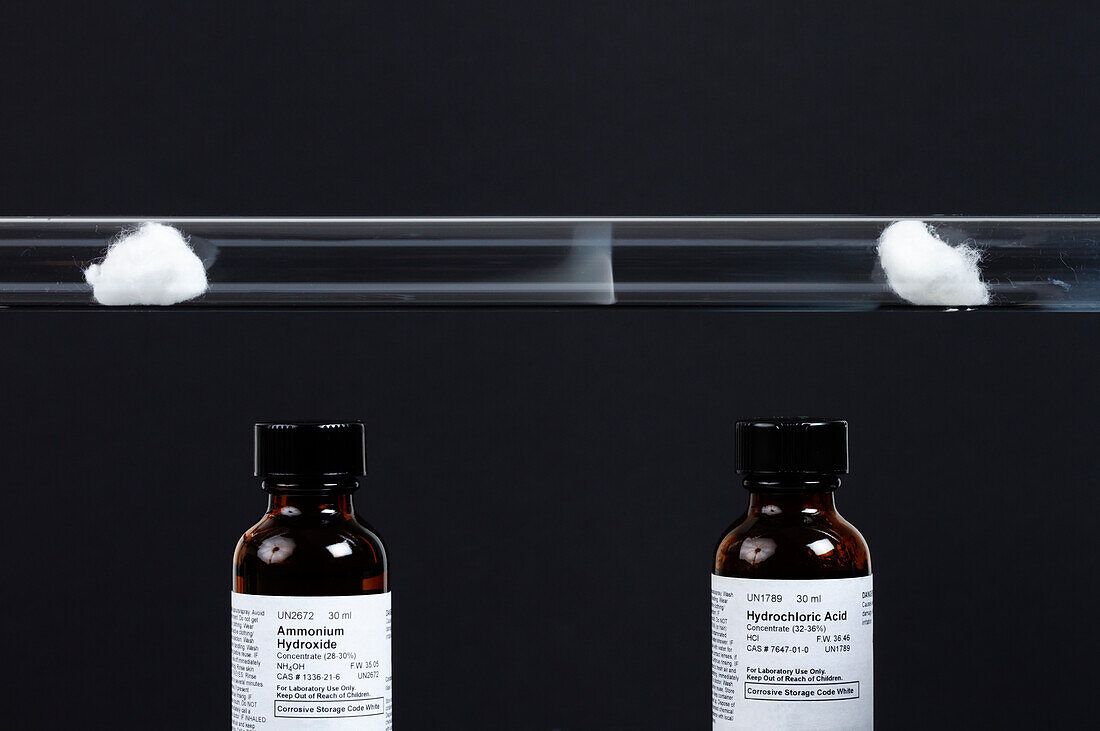
| Ammonium chloride formation. A glass tube is plugged by cotton balls at both ends. Small amounts of concentrated ammonium hydroxide (NH4OH, 28-30%) and concentrated hydrochloric acid (HCl, 32-36%) are used to wet the two balls. Colorless ammonia (NH3) and hydrogen chloride (HCl) gases diffuse from the respective balls. When they meet, they react and produce a ring of fine white powder of ammonium chloride: NH3 + HCl -> NH4Cl. Molecular weight of ammonia is less than that of hydrogen chloride, therefore it diffuses faster and the white ring is formed closer to the ball with the hydrochloric acid. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Turtle Rock Scientific |
| Taille de l’image : | 5160 px × 3429 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
