Aluminum reacts with hydrochloric acid, 5 of 6
Numéro d’image : 12649734
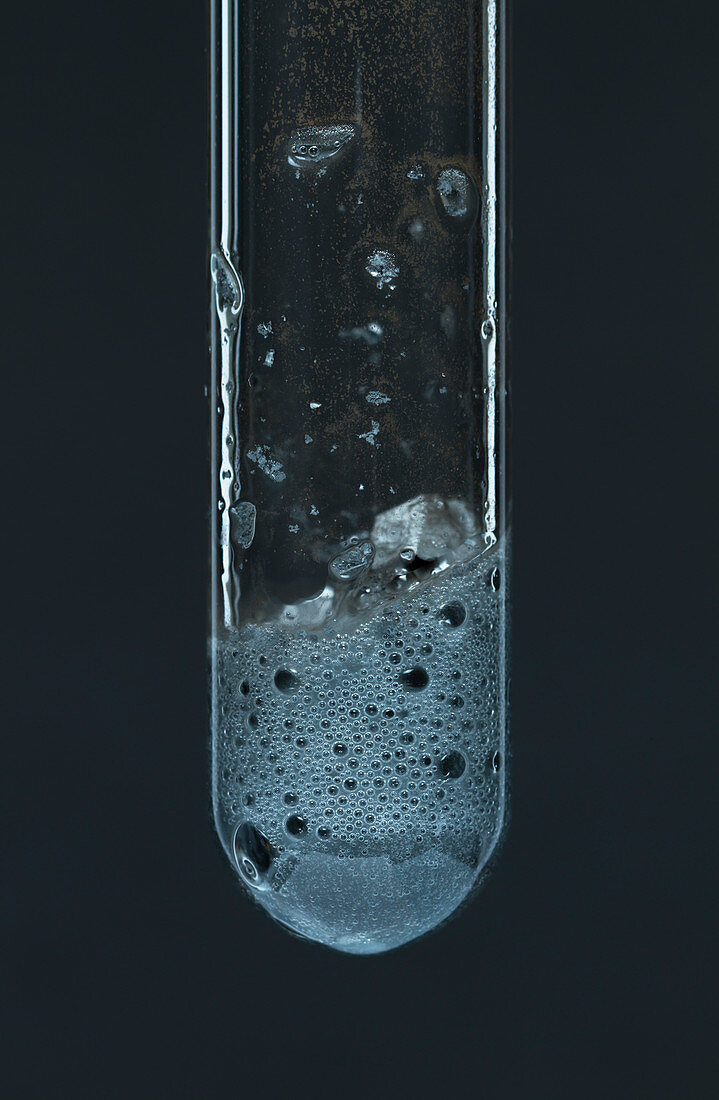
| Aluminum reacts with hydrochloric acid, 5 of 6. 1 millilitre of 5M hydrochloric acid (HCl) is added to crumbled aluminium metal (Al) foil in a test tube. Aluminum metal has a very thin but strong aluminium oxide (Al2O3) on its surface. For the first minute or so, there are no visible signs of a reaction as aluminium oxide is dissolved by the acid. After that hydrogen (H2) bubbles start to appear, temperature rises and the reaction speeds up greatly: Al + HCL -> AlCl3 + H2. As hydrochloric acid is used up, the reaction comes to a halt. This is an example of a single displacement reaction. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Science Source / Turtle Rock Scientific |
| Taille de l’image : | 3351 px × 5130 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
