Lithium Reacts with Water, 1 of 4
Numéro d’image : 12639484
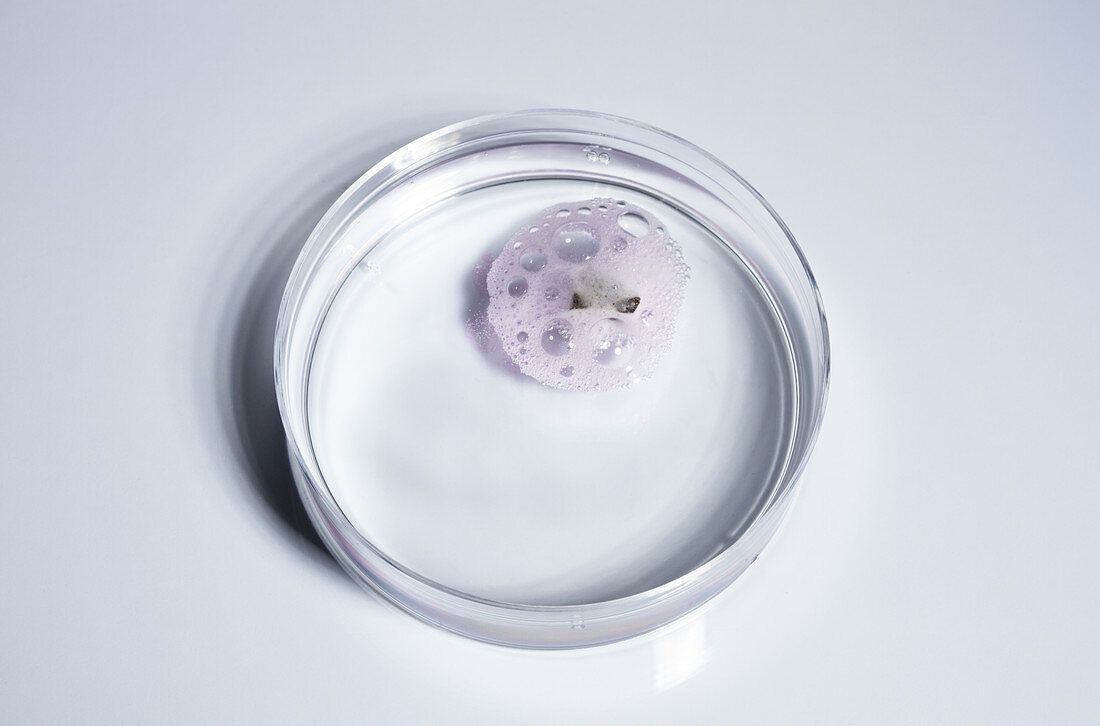
| Lithium reacts with water, 1 of 4. A small piece of lithium metal (Li) is dropped into a Petri dish with water (H2O). A drop of detergent was added to water to prevent the metal from sticking to the side of the dish. Lithium floats and fizzes, producing bubbles of hydrogen gas (H2) and lithium hydroxide (LiOH): Li + H2O -> LiOH + H2. The presence of the hydroxide is accentuated by the addition of few drops of phenolphthalein indicator, which turns the solution pink, indicating alkaline solution. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Turtle Rock Scientific |
| Taille de l’image : | 5144 px × 3395 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
