Lead chromate precipitate, 3 of 3
Numéro d’image : 12635657
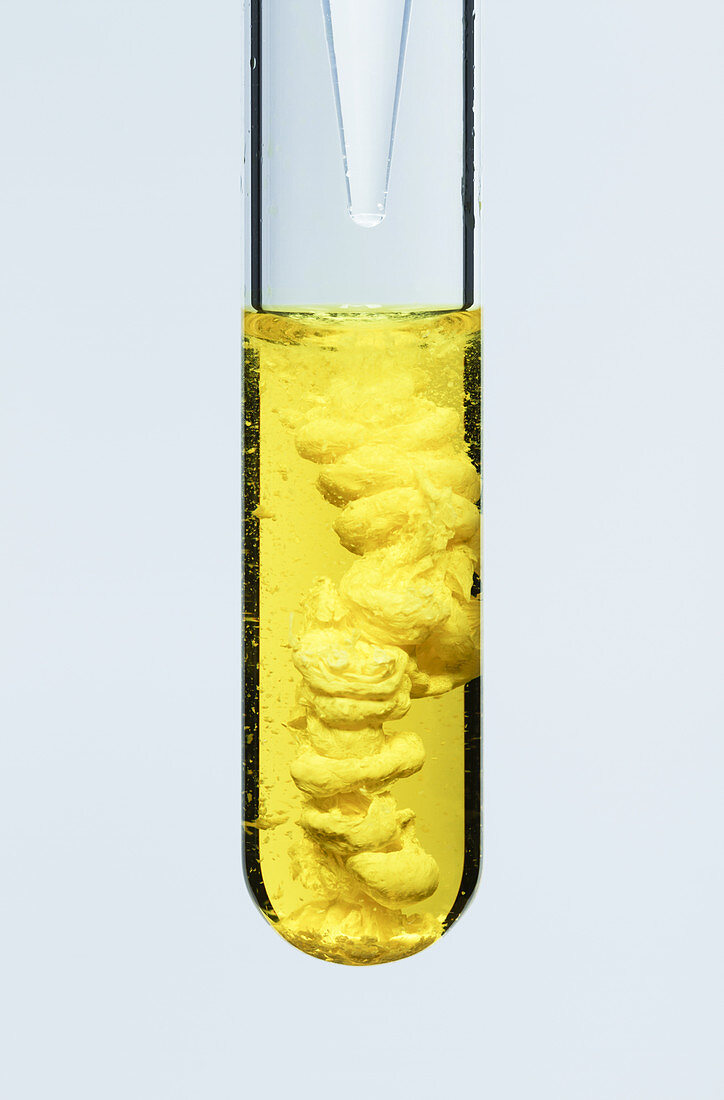
| Lead chromate precipitate, 3 of 3. Lead (II) chromate (PbCrO4) precipitate (bright yellow) formed by adding 0.25 M solution of lead nitrate (Pb (NO3)2) to 0.5 M solution of potassium chromate (K2CrO4). The reaction is K2CrO4 + Pb (NO3)2 -> PbCrO4 + KNO3. This is an example of a double replacement reaction. Lead chromate is the original chrome yellow paint pigment. | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Giphotostock |
| Taille de l’image : | 3395 px × 5157 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : | - |
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
Mots clés
- chimie,
- chimique,
- chromate,
- chromate de potassium,
- conduire,
- éprouvette,
- K2CRO4,
- métathèse,
- métathèse de sel,
- nitrate,
- PB NO32,
- potassium,
- précipitation,
- précipiter,
- réaction,
- réaction de déplacement,
- réaction de double déplacement,
- réaction de double remplacement,
- réaction de remplacement,
- sel,
- séquence,
- séries,
- solution
