Molecular graphic of graphite
Numéro d’image : 11997527
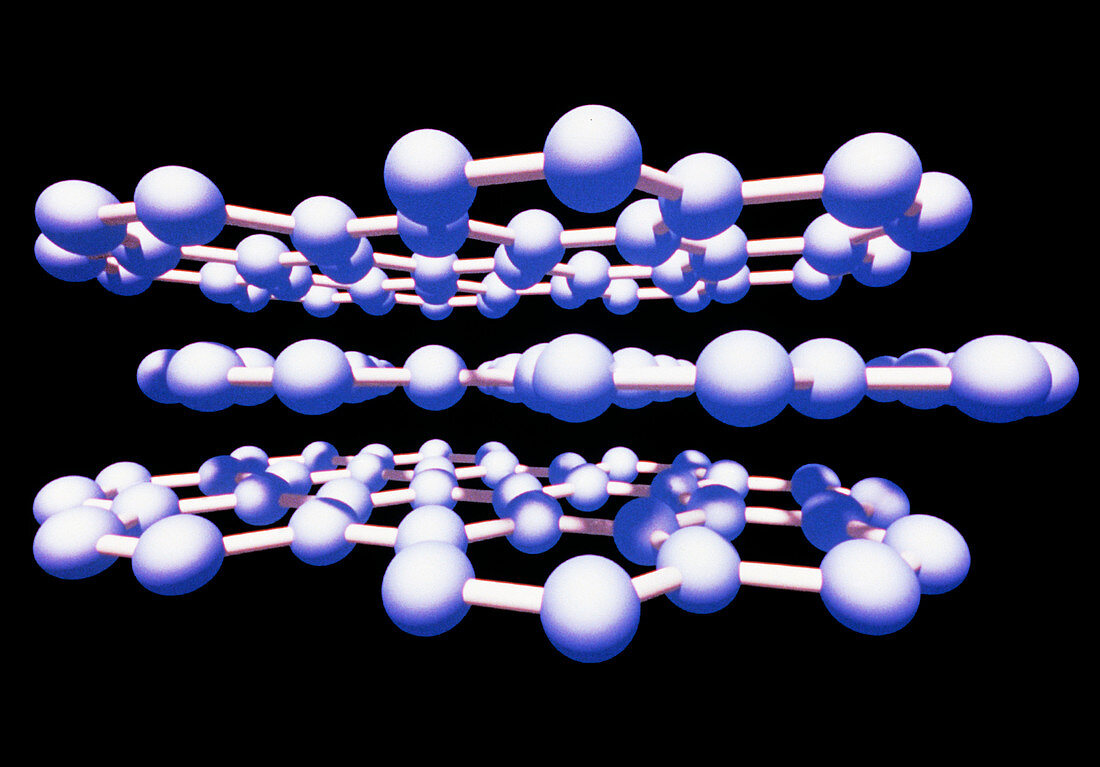
| Graphite. Molecular graphic of graphite,one of the naturally existing allotropes of the element carbon. Carbon atoms are represented by blue spheres. Graphite is used in pencil leads and as a lubricant. It has a crystalline structure composed of parallel layers of hexagonally arranged carbon atoms. Within each layer,carbon atoms are linked by strong aromatic bonds,while the parallel layers are held together by weak intermolecular forces called Van der Waals' forces. This Van der Waals's bonding is strong enough to hold the layers together,yet weak enough to allow them to slide over each other. This also confers graphite's softness and its ability to act as a lubricant | |
| Licence : | Droits gérés |
| Crédit: | Science Photo Library / Edward, Kenneth / Biografx |
| Taille de l’image : | 5083 px × 3543 px |
| Model Release : | Non requis |
| Property Release : | Non requis |
| Restrictions : |
|
Prix pour cette image À partir de 45 €
Produit vendu
(Calendrier, Carte postale, Carte de vœux, Impression sur textile, Packaging etc)
À partir de 45 €
Usage commercial
(Affichage, Annonce presse, Annonce TV, Carte, Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 45 €
Éditorial
(Digital, Journal, Livre, Livre pratique, Magazine, Télévision etc)
À partir de 60 €
Usage non-commercial
(Digital - hors rés. sociaux, Digital - rés. sociaux etc)
À partir de 120 €
